Unlocking the Secrets of Xist Regulation: A Journey Through CRISPR Innovation
Exploring the Role of Transcription Factors
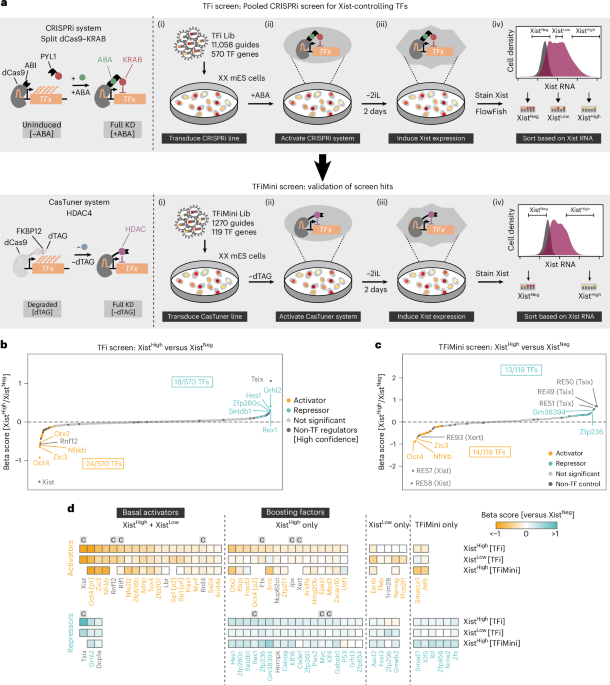
As I delve into the intricate world of gene regulation, one topic that continually captivates my attention is the process by which specific transcription factors (TFs) control gene expression, particularly in a gender-specific and developmental context. Recently, researchers employed pooled CRISPR interference (CRISPRi) screens to identify TFs that regulate Xist expression—a key player in X chromosome inactivation (XCI). This groundbreaking work not only sheds light on fundamental genetic mechanisms but also offers insights into potential therapeutic avenues for treating diseases linked to dysfunctional gene regulation.
The team conducted their investigation using differentiating mouse embryonic stem cells grown under conditions that mimic developmental trajectories seen in vivo. Utilizing a split dCas9–KRAB system allowed them to repress gene expression selectively, enabling a focused exploration of how various TFs impact Xist upregulation at the onset of XCI. The comprehensive CRISPR library comprised over 11,000 guide RNAs targeting more than 570 expressed TF genes—an ambitious undertaking that underscores our ever-deepening understanding of gene regulation.
“Notably, OCT4 emerged as a strong activator of Xist—contradicting its previous classification as a repressor.”
The Surprising Dynamics of OCT4
What struck me most about this research was the revelation regarding OCT4 (POU5F1)—a transcription factor typically cited as an inhibitor of Xist. Researchers found that knocking down OCT4 actually led to substantial upregulation of Xist in undifferentiated cells. It seems counterintuitive; how could a factor known for repression suddenly take on an activating role during differentiation? Perhaps it reflects the complexity and nuance inherent in cellular programming—a reminder that biology often defies straightforward categorizations.
I still wonder how many similar surprises await us as we explore other genes and pathways. Could there be hidden layers within our genetic blueprints waiting to be uncovered through innovative technologies like CRISPR? As scientists continue to unravel these complexities, we may start seeing profound implications for regenerative medicine and cancer therapy—fields where understanding cell behavior is crucial.
XCI Onset: An Intricate Dance
A significant aspect highlighted by this study involves how different groups of activators respond uniquely during various stages leading up to XCI. High-resolution RNA sequencing captured dynamic expression patterns across XX and XO cells during critical windows post-differentiation—a meticulous effort reminiscent of capturing fleeting moments through a camera lens. Early basal activators surfaced first, setting the stage for eventual high levels achieved by later-acting boosting factors.
“The early regulators ZIC3 and NFRKB predominantly affected proximal elements; meanwhile, late factors OTX2 and FOXD3 modulated distal regions.”
The Dual Nature of Activation
This duality fascinates me because it reflects broader themes about how technology informs our understanding not just at molecular levels but also at clinical fronts. In my experience within digital health ecosystems, I have often encountered an analogical relationship between early signals—like those from basal activators—and technological innovations’ groundwork laid before emerging tools transform workflows or patient outcomes.
The realization struck me: much like scientific discovery requires foundational understandings to build upon each successive layer effectively, so too does healthcare innovation necessitate iterative advancements before achieving seamless integration into practice.
Functional Interactions Revealed Through Reporter Screens
Taking a step further into addressing functional interactions among TFs and regulatory elements (REs), reporter screens provided another layer of insight into this complex regulatory web surrounding Xist expression. Generating reporter lines across various REs helped visualize their activity while elucidating interactions with identified TFs—essentially mapping out relationships that dictate when genes express or remain silent.
“High reporter activity appeared to increase screen sensitivity… suggesting potent Xist activators act partly through direct binding.”
A Glimpse Into Human Experience and Expression Control
This quest for clarity reminds me just how essential communication is—not only among scientists but also within healthcare systems striving for collaborative care models shaped around patient needs rather than institutional silos alone. We must navigate these complexities thoughtfully if we hope to unlock transformative therapies tailored specifically for individuals based on their unique genetic makeups.
Toward Understanding Distal Elements’ Role in Gene Silencing
I find myself reflecting deeply on one critical takeaway from this study—the necessity for effective communication between proximal REs and distal elements during processes like silencing via XCI initiation correlates directly with advantageous health outcomes across populations impacted by conditions stemming from faulty gene regulation processes both inherited or acquired over time.
“Silencing increases gradually with increased expression; there isn’t a strict threshold.”
The Implications Are Profound Yet Uneasy
This realization stirs conflicting emotions within me—while I feel awe toward humanity’s ingenuity pushing boundaries further every day through science… there lies discomfort too regarding ethical considerations around designer genes or potential misuse as capabilities expand rapidly beyond present-day checks or balances established thus far!